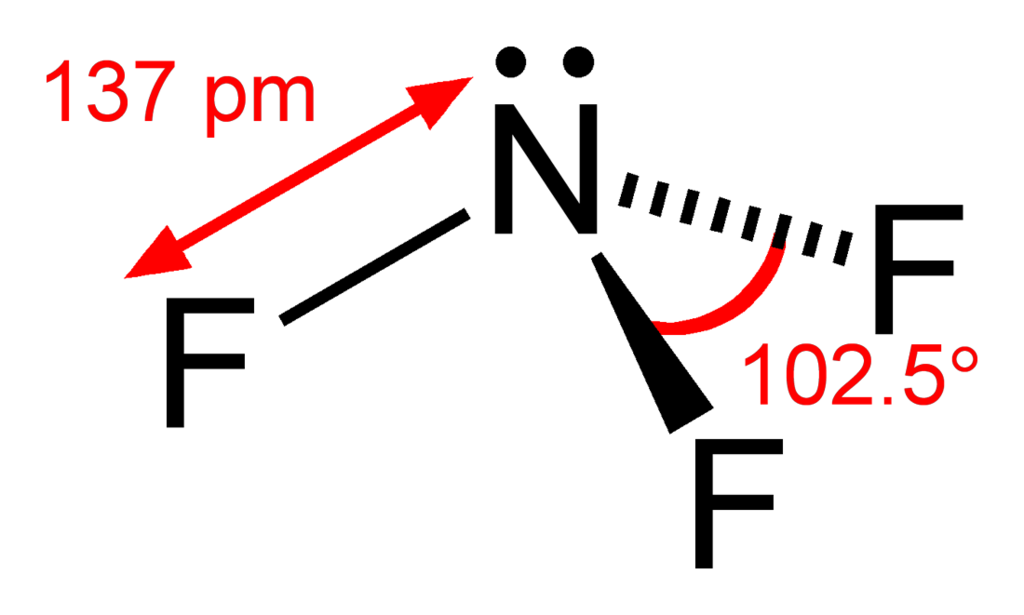
(a) Give the values for the indicated bond angles in the following molecules. 3 H H : H Br: (d) :Br Br:8 (b) Using the tables of electronegativities (EN) and the bond lengths from your SA tex.ġ8. of Molecule or Polyatomie lon Valence Electrons Lewis Structure Molecular Geometry Bond Angles Bond Polarity Molecular Dipole or Nonpolar Te amedral C.H.CI, H: 2 a = 14 109.5 lete polar S: triangular 107 S- SO, 410 nonpolar Botter Dobar 3270317 27 est (= 4 I ITO CHO Ic=0 bent 119 polar - 157 F-O-Ect 0= 6 bent 104.5 OF, F = 14 TOT polar 20 N=5 NO Linear 180 polar |0=12 anion Unear 1800 Nonpolar 1 - 0. of Molecule or Polyatomie lon Valence Electrons Lewis Structure Molecular Geometry Bond Angles Bond.ġ80 No. Explain what is wrong with each molecular geometry. Each ball-and-stick model shows the electron and molecular geometry of a generic molecule. How many double bonds are present in caffeine? How many single bonds? CH, HC CH 30. The structure of caffeine, present in coffee and many soft drinks is shown here. Determine the hybridization about Cin Hyco. Draw the correct Lewis structure for nitrogen trifluoride. Determine the electron geometry, molecular geometry, and idealized bond angles for PF3. 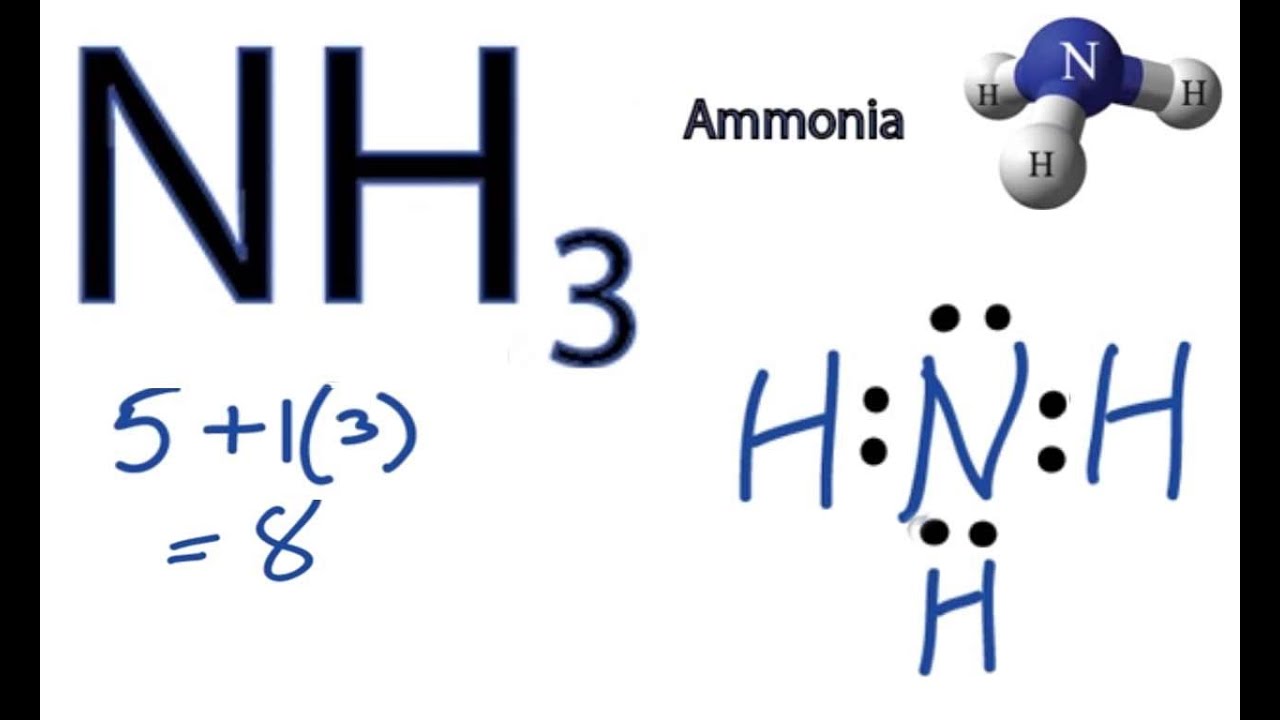
ġ25 Laboratory 9 Atomic and Molecular Structure Record the bond lengths and bond angles of H,O include units Bond Length он Bond Angle нон 0.990 A 164.51 What is the Electron-Domain Geometry of H,O? [1 point Tri gonal Planar What is the Molecular Shape of H,O? [1 point Tetiahedral What occupies the large portion of the VdW sphere away from the bonds in H,O? [1 point) Record the bond lengths and bond angles of NH include units. 125 Laboratory 9 Atomic and Molecular Structure Record the bond lengths and bond angles of H,O. 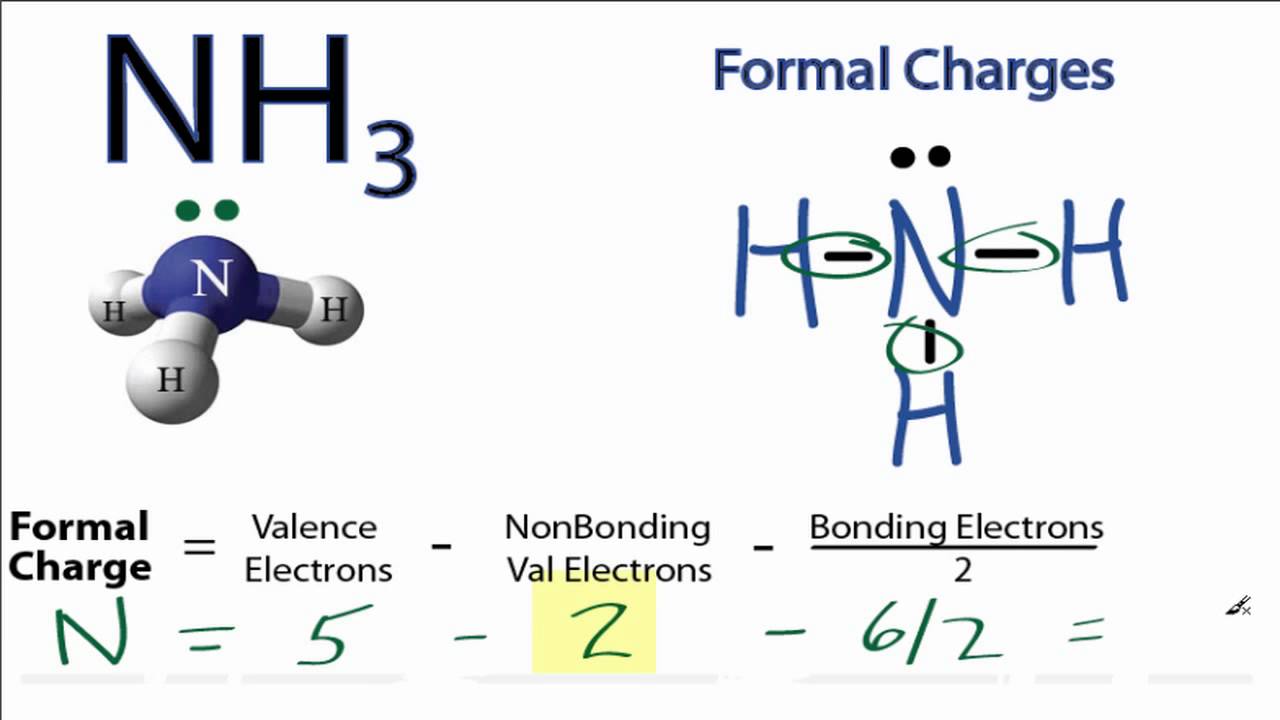
2) Using the informat ion at the top as a guide, what effects would replacing the phosphor i (plural for phosphorus) with arsenics have on the overa structure of DNA? 3) How are the molecular representation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
